Table of Contents
- About LPUNEST
- LPUNEST 2026 Exam Dates
- LPUNEST 2026 Application Form
- LPUNEST Exam 2026 Eligibility
- LPUNEST 2026 Admit Card
- LPUNEST 2026 Exam Centres
- LPUNEST 2026 Exam Pattern
- LPUNEST Entrance Exam 2026 Syllabus
- LPUNEST 2026 Reference Books
- LPUNEST Entrance Exam Preparation Tips
- LPUNEST Mock Test 2026
- LPUNEST 2026 Answer Key
- LPUNEST 2026 Results
- LPUNEST Courses 2026 Cut Off
- LPUNEST 2026 Counselling
- LPUNEST 2026 Scholarships
- LPUNEST 2026 Contact Details
The LPUNEST 2026 Exam registration began on 26th April 2026. The last date to apply for the exam and book their slot is "Three days before the conduct of the exam". The LPUNEST Result will be declared within 24 hours of the exam being conducted. The last date to get admission in B.Tech courses is May 20, 2026.
The LPUNEST 2026 result is released on the official website. The result is available for the candidates by logging into their accounts. The LPUNEST Result will be released in a scorecard form which will contain the information like Subject Wise Scores and Total Scores of the candidates. After the release of LPUNEST Result, the Candidates clearing the exam will be able to participate in LPUNEST Counselling 2026. The counselling schedule will be released soon after the release of result.
Lovely Professional University administers the National Entrance and Scholarship Test for admission to its various UG, PG and PhD programs. Every year, admission to more than 150 programs is performed online. The test serves as both an entrance test and a scholarship test for admission to the university.Interested candidates can check the article below to keep up with the latest events and notifications related to the LPUNEST exam.
About LPUNEST
The LPUNEST full form is the Lovely Professional University National Entrance and Scholarship Test. LPU is a private and one of the largest universities located in Jalandhar, Punjab, India, established in 2005 by Lovely International Trust under The Lovely Professional University Act, 2005. In addition, LPU received the highest NAAC (National Assessment and Accreditation Council) grade of 3.68 on a scale of 4.
LPU offers a scholarship for students aspiring to study in one of the courses offered at the university. It also holds approvals from the Punjab Pollution Control Board (PPCB) and Punjab State Power Corporation Limited (PSPCL) for its campus. The LPUNEST 2026 is conducted only online as a computer-based test (CBT). The LPUNEST 2026 is conducted only once a year, in two schedules.
The Lovely Professional University has several international tie-ups with many universities. A few are the University of Iowa, the University of East London, the University of Missouri, California State University, Otterbein University, Murdoch University, Jonkoping University, etc. In addition, it provides scholarships worth over 7.5 lakhs (for a 5-year course) for deserving candidates.
Courses Offered: LPUNEST 2026 is a university-level entrance exam inducted in the year 2009 for residential admissions into the Engineering, Management, Law, Design, Agriculture, Hotel Management & many other courses at Lovely Professional University.
|
Particulars |
Details |
|
Exam Name |
LPU NEST |
|
Full Form |
Lovely Professional University National Eligibility and Scholarship Test |
|
Conducting Body |
Lovely Professional University (LPU) |
|
Exam Level |
National Level Examination |
|
Courses Offered |
Undergraduate, Postgraduation and Ph.D. programs |
|
Exam Mode |
Online Mode (Remotely Proctored Test) |
|
Type of Questions |
Multiple Choice Questions |
|
Official Website |
|
|
Helpdesk |
Tel: +91-1824-404404 Toll-Free: 1800 102 4431 Toll-Free: 1800 274 0615 |
LPUNEST 2026 Courses
Apart from conventional engineering streams, LPUNEST 2026 also offers a number of trend-setting streams/ majors for B.Tech courses. The various streams offered for the B.Tech course are:
- Electronics and Communication Engineering (ECE)
- Computer Science and Engineering (CSE)
- Information Technology (IT)
- Mechanical Engineering (ME)
- ME - Mechatronics
- Aerospace Engineering
- Automobile Engineering (AE)
- Civil Engineering (CE)
- Electrical Engineering (EE)
- Electrical and Electronics Engineering (EEE)
- Biotechnology (BT)
- Food Technology
- Biomedical Engineering
- Chemical Engineering (CHE)
- CHE - Petroleum
LPUNEST 2026 Exam Dates
It has many courses for which the detailed LPUNEST 2026 Exam Dates are below:
LPUNEST 2026 Exam Dates for B.Tech
|
Particulars
|
BTECH & Other test |
| Phase 2 | |
| Online Registration Starts on | 26th April 2026 |
| Slot Booking for Online Examination | 3rd May 2026 onwards |
| Last date to apply & book slot | 15th May 2026 |
| Dates of Entrance Examination # | 4th May onwards |
| Display of Result | Within 24 working hours |
| Last Date of Admission | 20th May 2026 |
LPUNEST 2026 Exam Dates for MBA
| Type B interview programmes (MBA) |
|
| Particulars | Phase 2 |
| Online Registration Starts on | 26th April 2026 |
| Slot Booking for Online Examination | 3rd May 2026 onwards |
| Last date to apply & book slot | 15th May 2026 |
| Dates of Entrance Examination | 4th May onwards |
| Last date to upload Video Essay | Exam date + 1 day For Example : If candidate has appeared for exam on 23rd April, then last date to upload Video essay is 24th April. |
| Display of Result | Within 24 working hours |
| Slot booking for Interview |
Candidate can opt. for any preferred interview slot within 2-7 days after the declaration of LPUNEST result. For Example: If result is declared on 5th May 2026, Interview can be scheduled on any day between 7th May to 12th May 2026.
|
| Dates of Interview | |
| Result for Interview | Within 24 working hours |
| Last Date of Admission | 20th May 2026 |
LPUNEST 2026 Exam Dates for Design
|
Particulars
|
Type B (UG Design) |
| Phase 2 | |
| Online Registration Starts on | 26th April 2026 |
| Slot Booking for Online Examination | 3rd May 2026 onwards |
| Last date to apply & book slot | 15th May 2026 |
| Dates of Entrance Examination # | 4th May onwards |
| Display of Result | Within 48 working hours |
| Last Date of Admission | 20th May 2026 |
Also Read: LPU Grading System 2026
LPUNEST 2026 Application Form
LPUNEST 2026 application form is available online.The last date to submit the LPUNEST application form is 3 days prior to the exam for the B.Tech courses.
Candidates must note that the last date of slot booking varies for different courses, and therefore, they must check out the detailed exam schedule released by the conducting body.
Once the candidates have registered and filled out the LPUNEST application form 2026 details, they will have to pay the application fee. Candidates can pay the fee online only through net banking/credit card/debit card.
Candidates without the online internet facility can get the application form (and the LPUNEST Kit) by visiting the University Campus or any other prescribed place by paying cash.
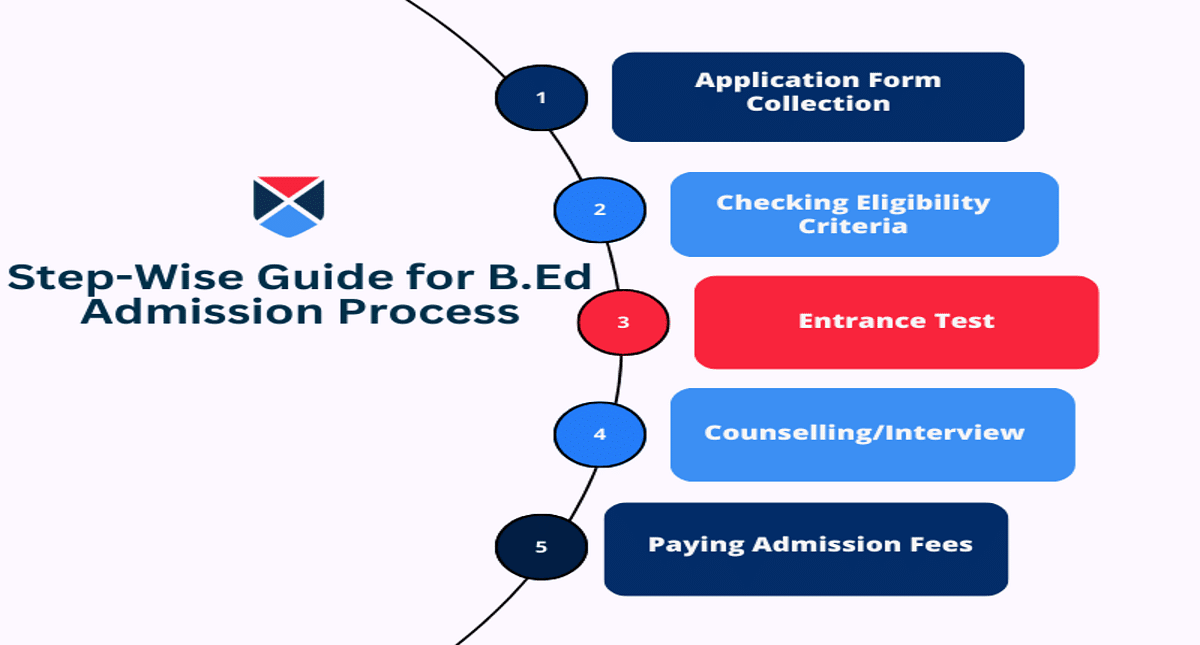
LPUNEST Entrance Exam 2026 Application Process
Lovely Professional University has released the application on its official website. Candidates must fill in the LPUNEST 2026 application form details and proofread it before submitting it. Aspirants can go through the following sections to get familiarised with the application process.
Applicants should understand the LPUNEST application process carefully. Candidates can follow these steps to apply for the LPUNEST 2026 exam.
Step 1: Registration
- Candidates can register by visiting the official website.
- Provide all necessary details such as names, email IDs, date of birth, and valid mobile number after which a registration ID will be generated and sent to their registered email ID.
Step 2: Payment of Application Fee
- After successful registration for LPUNEST 2026 candidates can proceed with the payment of the application fee. The modes of paying the application fee are online using net banking, credit, or debit card.
Step 3: Online Application Form Filling
- Post completion of the application fee payment, candidates must fill in all personal, academic, and contact details in the application form. Candidates must preview all details provided in the application form. In case the form is incomplete or incorrect then the application will be rejected.
Step 4: Uploading Scanned Documents
- Candidates can proceed to the next steps which are to upload the scanned images of the documents after filling in the details in the application form.
- Candidates should upload scanned copies of the photograph, birth certificate/Aadhaar card, and certificate of the qualifying exam as per the specifications mentioned by the university.
- The candidate should have a recent photo taken against a white background. The size of the photo should be 10 KB to 100 KB. Candidates should have a scanned copy of their signature. They have to remember that the signature should be consistent throughout the admission process else they will be subjected to rejection.
Step 5: Submission of Application Form
- After successfully uploading the required documents, candidates have to finally submit the application form for LPUNEST 2026 by clicking on a button given at the last of the form.
- Candidates must print a copy of the filled application form for future reference
LPUNEST Application Form 2026 Fee
The fee which has to be paid for a successful application is as follows.
|
Category |
Cost of Application Form |
|
Male |
INR 1000 |
|
Female |
INR 500 |
Documents Required on LPUNEST 2026 Application Form
The official authorities at LPU require certain documents for verification purposes. The various documents that candidates require while applying for the LPUNEST 2026 exam are mentioned below.
- Marksheet of Class 10th and 12th
- Valid Mobile Number and Email ID
- Payment options like Debit card/Credit card/Net banking/Demand Draft
- Scanned copy of the candidate's signature
- A recent passport-size photograph against a white background
LPUNEST Exam 2026 Eligibility
The Lovely Professional University offers admission into various undergraduate and postgraduate courses. Therefore, the eligibility of candidates depends on the course for which they are applying. Only Indian Nationals are eligible to apply for the LPUNEST 2026 exam.
LPUNEST 2026 Eligibility for Various UG Courses
Applicants should have either completed or should be appearing in 2026 for the final examination of 10+2 from the Central Board of Secondary Education (CBSE, New Delhi) or they should be appearing for the Council for Indian School Certificate Examination (CISCE, New Delhi) or State Board or any other Institution, college. These institutions must be recognised by the Council of Boards of School Education (COBSE), New Delhi, or any other concerned apex body as applicable.
LPUNEST 2026 Eligibility for B.Tech
The qualifying candidates must have secured minimum aggregate marks of 60% in 10+2 or equivalent (with Physics, Mathematics, and English), subject to qualifying for LPUNEST (B.Tech). There is 5% relaxation to North-East states and Sikkim candidates/ Defence Personnel and their Dependents/ Wards of Kashmiri Migrants as far as passing marks in the qualifying exam are concerned.
There is a 10% relaxation to outstanding Sports Persons (i.e. A/A+ category as per Sports Scholarship) as far as passing marks in the qualifying exam are concerned, Subject to verification from the selection committee of the University. The selection committee must comprise the Head (DSW), Director of Sports, Deputy Director of Sports, Concerned Coach, and VC Nominee.
LPUNEST 2026 Admit Card
LPU, the conducting authority of LPUNEST, will release the admit card on the official website within 2 days of completing the online slot booking process.
Candidates must keep their admit cards in their possession throughout the admission process. They can download their admit cards by entering their application number and password. A copy of the LPUNEST 2026 admit card is mailed to the candidate's mail ID provided during registration.
How to Download LPUNEST Admit Card?
The official authorities at LPU release admit cards on the university website. Once the admit card is released, candidates can download it by following the steps below:
- Step 1: Candidates must visit the official website of LPUNEST 2026.
- Step 2: Click on the "LPUNEST" icon.
- Step 3: Log in using your credentials, which are User ID and Password.
- Step 4: Click on submit. LPUNEST 2026 admit card will be displayed on the screen.
- Step 5: Download and take a printout of the Admit Card on an A4-sized sheet.
Details on LPUNEST Admit Card
Details mentioned on the LPUNEST admit card are very important. Applicants should carefully go through the details mentioned on the LPUNEST 2026 admit card. Candidates can find the details on the LPUNEST 2026 admit card.
- Candidate's Name
- Roll number
- Date of Birth
- Candidate's photograph
- Test centre address
- Exam date and Slot time
- Course Name
- Exam instructions
LPUNEST 2026 Exam Centres
Exam centres are spread across 200 cities with nearly 140 exam centres. The candidates can select the exam centre at the time of filling out the LPUNEST application form. The candidate can choose the centre which is comfortable and convenient. LPU NEST 2026 can be attended either in English or Hindi.
The candidates are advised to get detailed knowledge about LPUNEST Centres before filling out the application form. This will be beneficial for them in spotting the centre closest to their location.
Read More: LPUNEST Exam Centres
LPUNEST 2026 Exam Pattern
Each course's LPUNEST 2026 exam pattern and question paper is unique. In the LPUNEST Question Paper, there will be four sections: Physics, Chemistry, Mathematics/Biology, and English. Each section accounts for 33% of the total weightage of the paper.
Refer to the table below to learn more about the LPUNEST 2026 exam pattern:
|
Particulars |
Details |
|
Mode |
Online |
|
Duration of Examination |
2 Hours 30 Minutes |
|
Medium of Language |
English |
|
Sections |
Physics - 30 Questions Chemistry - 30 Questions Mathematics/Biology - 30 Questions English - 30 Questions |
|
Nature of Question |
Objective i.e., Multiple Choice Questions |
|
Total Number of Questions |
120 Questions |
|
Marking Scheme |
4 marks will be awarded for PCM/B sections for each correct answer. 2 marks will be awarded for the English section for each correct answer. No marks will be deducted for wrong responses or unanswered questions |
Also Read: How to Crack LPUNEST?
LPUNEST Entrance Exam 2026 Syllabus
The LPUNEST is an exam that is conducted to offer admissions into various undergraduate and postgraduate courses. Therefore, the LPUNEST syllabus 2026 that a candidate should concentrate on depends on the course that he/she is applying for.
Applicants are advised to go through the course syllabus carefully and cover all the mentioned topics during the preparation process.
- Syllabus for B.Tech: Topics are related to subjects like Physics, Chemistry, Mathematics/Biology, and English. Candidates must go through the 10th and 12th class syllabi of these subjects to prepare well for the exam.
- Previous Year Paper/Sample Paper: Candidates can refer to the previous year's question papers to get a clear idea of the syllabus, exam pattern, and marking scheme.
- Other Courses: If a candidate is applying for other courses, then the syllabus will be related to the course for which they are applying. The LPUNEST Syllabus will also depend on the qualifying or qualifying exam.
LPUNEST 2026 concentrates most on the students' strengths in the 11th and 12th syllabi. It puts to test the subject understanding of the candidates on how well he/she understand the studied or given syllabus. Candidates can find the LPUNEST syllabus here.
LPUNEST 2026 Reference Books
Candidates aspiring to take admission to Lovely Professional University (LPU) must learn and practice questions from good reference books. Since there are many courses under LPUNEST, each course has its own syllabus and reference books. The list of reference books for candidates preparing for LPUNEST 2026 is given below.
- NCERT Textbook for Class 11th and 12th (PCMB)
- Concepts of Physics(Volume 1 and 2) by HC Verma
- RD Sharma's Mathematics classes 11th and 12
- Concise Inorganic Chemistry by JD Lee
- Universal's CLAT Guide
- Legal Awareness and Legal Aptitude by AP Bhardwaj
- NIFT, NID & IIFT Entrance Exam Guide By D Mittal
- General Aptitude: Quantitative Aptitude & Reasoning
- General Aptitude Theory and Practice by Anand Kumar and Ajay Kumar
- How To Prepare For CAT by Arun Sharma (All 3 Books - VARC/DILR/QA)
LPUNEST Entrance Exam Preparation Tips
To prepare well for the exam and to score good marks, candidates must have a well-planned schedule. Candidates can refer to the preparation strategies to be followed before the exam. Some of these are listed below:
- Make a proper schedule
- Must have an in-depth knowledge of the exam syllabus and pattern.
- Solve previous years' question papers to know the pattern of question papers.
- Attempt mock tests to manage time effectively.
- Make short notes for the important topics.
- Do a quick revision before the exam and study at least 8-10 hours per day.
Also Read: Reasons to Give LPUNEST Exam
LPUNEST Mock Test 2026
LPU provides mock tests on its official website to help candidates study for the LPU NEST exam. A mock test is a practice version of the actual exam that allows students to familiarise themselves with the format and difficulty level of the exam.
Candidates should take the sample test to familiarise themselves with the exam structure and identify areas where they need to improve. This can help students perform better on the LPU NEST exam.
Refer to the table below to take the LPUNEST mock test:
|
LPUNEST Mock Test Link |
LPUNEST 2026 Answer Key
To download the LPUNEST 2026 answer key, candidates must visit the official website of Lovely Professional University. The answer key release dates are yet to be announced by the conducting authorities.
The following are the steps to download the LPUNEST answer key 2026:
- Visit the official website of LPU.
- From the homepage, navigate to the "Answer Key" tab.
- The LPUNEST answer key will be displayed on the screen.
- Candidates must cross-verify their responses with the answer key and mark themselves with the help of the LUPNEST marking scheme.
- Save the answer key for further processes.
LPUNEST 2026 Results
LPUNEST results 2026 for Phase 4 will be declared within 48 working hours after the exam. LPUNEST result is announced on the Lovely Professional University official website in PDF format.
Candidates can take a printout for future reference. In addition, candidates can check the LPU results by following the steps below:
Steps to Check LPUNEST 2026 Results
The LPUNEST result is announced on the official website by the conducting body of the exam. Applicants can check the results by following the steps mentioned below:
- Step 1: Candidates must visit the official website of LPUNEST 2026 and get to the login page.
- Step 2: Enter the valid credentials such as email ID/mobile number/application number and password. Click on the 'Login' button.
- Step 3: Click on the 'View Results' tab, and view LPUNEST 2026 results.
- Step 4: Take a printout of the LPUNEST 2026 result for future reference.
Details Mentioned on LPUNEST 2026 Scorecard
Applicants should carefully check the details mentioned on the LPUNEST scorecard. For example, the scorecard of LPUNEST contains the following information.
- Candidate's Name
- Roll Number
- Category
- Course Name
- Composite Score
- Sectional Score
- Percentile
- Overall Marks Obtained.
LPUNEST Courses 2026 Cut Off
LPUNEST Cut Off 2026 is the minimum score that the candidate must score to be eligible for admission to the Lovely Professional University National Entrance and Scholarship Test.
LPUNEST Cut-Off 2026 marks for B.Tech courses can be judged based on the previous year's cutoffs. Candidates will also be the scholarships that score more than or equal to the cutoff marks.
Factors Determining LPUNEST Courses Cut Off 2026
The following factors are considered while evaluating the LPUNEST cutoff.
- The minimum marks are set by the University and are based on a number of factors.
- The number of candidates, number of vacant seats, average performance in the entrance test, and last year's cut off.
- After the cut-off is declared, a Merit List will be prepared on the basis of the candidate's scores on the entrance test.
LPUNEST 2026 Expected Cut Off
The table below represents the expected LPUNES 2026 cut off.
|
Cut Off Category |
Marks required out to 420 |
|
Category I |
320 |
|
Category II |
240 |
|
Category III |
165 |
Also Read: B.Tech Entrance Exams
LPUNEST 2026 Counselling
LPUNEST 2026 counselling will commence after the result is declared by the conducting body. The counselling and admission process of Lovely Professional University (LPU) will be conducted online.
Based on the merit list, the candidates will be called for the LPUNEST counselling process. Allotment of the seats is based on the candidate's ranking in the exam, preferences, and availability of the seats.
Documents Required for LPUNEST 2026 Counselling
Candidates are required to bring the following documents at the time of the LPUNEST 2026 counselling process:
- 10th Marksheet
- 12th Marksheet and Pass Certificate
- LPUNEST 2026 Admit Card
- LPUNEST 2026 Scorecard
- Any Photo identity or Government ID proof such as Aadhar, PAN card, Driving License, Passport etc.
- Caste Certificate (If any)
- Birth Certificate
- Category Certificate
- Character Certificate
- Passport size photograph
LPUNEST 2026 Scholarships
LPUNEST 2026 is both an admission exam and a scholarship test. LPU awards scholarships depending on a variety of variables. Scholarships are offered to eligible individuals depending on their performance in any of the following categories:
Qualification for LPUNEST Scholarship Eligibility
LPU's Entrance and Scholarship Test: LPUNEST National level exams including JEE Mains, NEET, UCEED, CAT, MAT, GATE, GPAT, and others.
- Scholarship Scholarship brackets are set for all of the categories indicated above (Bracket 1, 2, and 3).
- Each bracket has fixed requirements (qualifying exam results) that candidates must meet to be placed in that bracket.
- The criterion in each bracket will be lowered as the process progresses to the next rounds, as will the scholarship amount.
- As a result, students who fall into the Bracket-1 criteria will receive the highest scholarship compared to those who fall into Brackets 2 or 3.
|
Phases |
Bracket-1 |
Bracket-2 |
Bracket-3 |
|
Phase I |
50% of the course Fee |
40% of the course Fee |
30% of the course Fee |
|
Phase II |
45% of the course Fee |
35% of the course Fee |
25% of the course Fee |
|
Phase III |
40% of the course Fee |
30% of the course Fee |
20% of the course Fee |
|
Phase IV |
35% of the course Fee |
25% of the course Fee |
20% of the course Fee |
LPUNEST 2026 Contact Details
For the LPUNEST entrance exam 2026 and admission-related queries, candidates can contact the address and phone number provided below:
AddressLovely Professional University, Jalandhar - Delhi G.T. Road, Phagwara, Punjab (India) - 144411.
Contact Number: +911824517000, +911824404404
Email: admissions@lpu.co.in (Admissions), int.admissions@lpu.co.in (International Admissions)
Official Website: www.nest.lpu.in/main.aspx
FAQs on LPUNEST
Q: What are the courses offered through LPUNEST 2026?
LPUNEST offers courses from various streams such as Engineering, Management, Law, Design, Agriculture, Hotel Management, Pharmacy, Medical, and much more.
Q: What is the Application Fee for LPUNEST 2026?
The application fee for the LPUNEST kit and the application form is as follows: Cost of Application Form and Soft Copy of LPUNEST Kit For Boys - INR 1000 For Girls - INR 500 Cost of Application Form and Printed LPUNEST Kit For Boys - INR 1500 For Girls - INR 1000
Q: How can I retrieve back my login credentials in case I forget?
If a candidate forgets their login password, they can click on forgot password link on the official website and enter their registered email id to retrieve back the password.
Q: What are the eligibility criteria for B.Tech through LPUNEST?
The candidates must have passed the 10+2 examination with Physics, Mathematics, and English as a mandatory subject with a minimum of 60% marks from a recognized board of education.
Q: On what basis is the cut off calculated in LPUNEST 2026?
The cut off will be calculated based on the Number of Candidates, Number of Vacant Seats, Average Performance LPUNEST, and Last year's cut off. Candidates must prepare well to qualify and participate in the counselling process.
Q: What is the counselling process of LPUNEST 2026?
The counselling for LPUNEST 2026 will be conducted online. At the first stage, the candidate documents will be verified and then the preferred seats will be allotted based on the marks secured in the qualifying examination based on category.
Q: If candidates is expecting 60% marks. Is there any chance of availing scholarship?
Scholarship is given to all deserve candidates based on various factors such as the scores, co-curricular, sports and national level exams such as JEE Mains, GATE, CAT, MAT, GPAT and more.
Q: What are the LPUNEST 2026 dates for registration and slot booking for B.Tech programs?
LPUNEST exam registration and slot booking for B.Tech admission program commenced on Oct 15, 2024. The last date for registration is Jan 15, 2026. The slot booking began on Dec 10, 2024, and will end at the same time as the application.