Table of Contents
AP EAMCET syllabus 2025 was released by JNTU on their official website, along with the information brochure of AP EAMCET 2025. AP EAMCET syllabus 2025 consists of topics from three sections - Mathematics, Physics, and Chemistry. The syllabus link of AP EAMCET for BIPC students 2025 was also shared on the official website.
Aspirants need to check the AP EAMCET 2025 syllabus for an effective AP EAMCET preparation 2025. The exam will be conducted in two sessions, comprising 160 questions each. Candidates are advised to go through the detailed syllabus to ensure that they do not miss any important topics and attempt a maximum number of questions in the test.
Must Practice: AP EAMCET Mock Test
Subject-wise AP EAMCET Syllabus PDF 2025
The AP EAMCET 2025 syllabus was shared by the official authority on the website. Candidates were provided with the syllabus PDF links for each course. Checking the the AP EAMCET syllabus 2025 comprehensively can help candidates prepare for the upcoming exam in a very organised and effective manner.
Candidates can find the subject-wise AP EAMCET syllabus 2025 in the form of PDFs from the table provided below:
| Subjects | Download PDF Link |
| Physics | Download here |
| Chemistry | Download here |
| Mathematics | Download here |
AP EAMCET 2025 Syllabus Important Topics
Here is a compilation of the AP EAMCET Syllabus 2025 according to the subject-wise list of all chapters covered. Candidates must ensure that they allocate the necessary preparation time for all of the concepts that are present in the exam.
Also, they have to practice answering the questions as fast as possible because you should be able to answer 160 questions in 180 minutes.
1. Physics
- Stoichiometry.
- Physical-World.
- Units and Measurement
- Motion in a Straight Line
- Motion in a Plane
- Laws of Motion
- Work, Energy and Power
- Systems of Particles and Rotational Motion
- Oscillations
- Gravitation
- Mechanical Properties of Solids
- Mechanical Properties of Liquids
- Thermal Properties of Matter
- Kinetic Theory
- Waves
- Ray Optics and Optical Instruments
- Wave Optics
- Electric Charges and Fields
- Electrostatic Potential and Capacitance
- Current Electricity
- Moving Charges and Magnetism
- Magnetism and Matter
- Electromagnetic Induction
- Alternating Current
- Electromagnetic Waves
- Dual Nature of Radiation and Matter
- Atoms
- Nuclei
- Semiconductors Electronics: Materials, Devices, and Simple Circuits
- Communication Systems
2. Maths
- ALGEBRA
- Trigonometry
- Vector Algebra
- Coordinate Geometry
- Calculus
- Probability
3. Chemistry
- Classification of Elements and Periodicity in Properties
- Chemical Bonding And Molecular Structure
- States of Matter: Gases and Liquids
- Thermodynamics
- Chemical Equilibrium and Acids-Bases
- Hydrogen and its Compound
- S - Block Elements (Alkali and Alkaline Earth Metals)
- P- Block Elements Group 13 (Boron Family)
- P-Block Elements -Group 14 (Carbon Family)
- Environmental Chemistry
- Organic Chemistry-Some Basic Principles and Techniques and Hydrocarbons
- Solid State
- Solutions
- Electrochemistry and Chemical Kinetics
- Surface Chemistry
- General Principles of Metallurgy
- P-Block Elements
- D and F Block Elements & Coordination Compounds
- Polymers
- Biomolecules
- Chemistry in Everyday Life
- Haloalkanes and Haloarenes
- Organic Compounds Containing C, H, & O like Alcohols, Phenols, Ethers, Aldehydes, Ketones and Carboxylic Acids
- Organic Compounds Containing Nitrogen
4. Botany
- Diversity in the Living World
- Structural Organization in Plants-Morphology
- Reproduction in Plants
- Plant Systematics
- Cell Structures and Functions
- Internal Organization in Plants
- Plant Ecology
- Plant Physiology
- Micro-Biology
- Genetics
- Molecular Biology
- Biotechnology
- Plants, Microbes and Human Welfare
5. Zoology
- Body Fluids and Circulation
- Human Reproductive System
- Study of Periplaneta Americana (Cockroach)
- Zoology-Diversity of the Living World
- Digestion and Absorption
- Breathing and Respiration
- Muscular and Skeletal System
- Organic Evolution
- Immune System
- Locomotion and Reproduction in Protozoa
- Excretory Health & their Elimination
- Reproductive health, Genetics & Applied Biology
AP EAMCET 2025 B.Tech Syllabus (Unit-wise)
AP EAMCET Syllabus MPC 2025 includes topics from Classes 11 and 12. Candidates can prepare for the exam using the AP EAMCET 2025 Reference Books, which will also contain some model questions and test papers. The unit-wise syllabus for Chemistry, Physics, Mathematics, and Biology is given below.
AP EAMCET 2025 Chemistry Syllabus
The chemistry syllabus for AP EAMCET has been listed below in a very detailed manner.
Atomic Structure
- Subatomic Particles
- Atomic Models –Rutherford’s Nuclear Model of an Atom
- Developments to the Bohr’s Model of an Atom
- Nature of Electromagnetic Radiation
- Particle Nature of Electromagnetic Radiation- Planck’s Quantum Theory
- Bohr’s Model for Hydrogen Atom Explanation of the Line Spectrum of Hydrogen
- Limitations of Bohr’s Model
- Quantum Mechanical Considerations of Subatomic Particles
- Dual Behaviour of Matter
- Heisenberg’s Uncertainty Principle
- Quantum Mechanical Model of an Atom
- Important Features of the Quantum Mechanical Model of an Atom
- Orbitals and Quantum Numbers Shapes of Atomic Orbitals
- Energies of Orbitals
- Filling of Orbitals in Atoms
- Aufbau Principle,
- Pauli’s Exclusion Principle and Hund’s Rule of Maximum Multiplicity
- Electronic Configurations of Atoms
- Stability of Half-Filled and Filled Orbitals
Classification Of Elements And Periodicity In Properties
- Need to Classify Elements, Genesis of Periodic Classification
- Modern Periodic Law and the Present Form of the Periodic Table
- Nomenclature of Elements with Atomic Number Greater than 100
- Electronic Configuration of Elements and the Periodic Table
- Electronic Configuration and Types of Elements s,p,d and f Blocks
- Trends in Physical Properties:(a) Atomic Radius, (b) Ionic Radius (c)Variation of Size in Inner Transition Elements, (d) Ionization Enthalpy, (e) Electron Gain Enthalpy, (f) Electronegativity
- Periodic Trends in Chemical Properties:(a) Valence or Oxidation States, (b) Anomalous Properties of the Second-Period Elements - Diagonal Relationship Periodic Trends and Chemical Reactivity
Chemical Bonding And Molecular Structure
- Kossel - Lewis Approach to Chemical Bonding, Octet Rule, Representation of Simple Molecules, Formal Charges, Limitations of the Octet Rule
- Ionic or Electrovalent Bond - Factors Favourable for the Formation of the Ionic Compounds-Crystal Structure of Sodium Chloride, General Properties of Ionic Compounds
- Bond Parameters - Bond Length, Bond Angle, and Bond Enthalpy, Bond Order, Resonance-Polarity of Bonds Dipole Moment-Fajan Rules
- Valence Shell Electron Pair Repulsion (VSEPR) Theory Predicting the Geometry of Simple Molecules
- Valence Bond Theory-Orbital Overlap Concept-Directional Properties of Bonds-Overlapping of Atomic Orbitals-Types of Overlapping and Nature of Covalent Bonds-Strength of Sigma and Pi Bonds-Factors Favouring the Formation of Covalent Bonds
- Hybridization- Different Types of Hybridization Involving s, p and d Orbitals- Shapes of Simple Covalent Molecules Coordinate Bond -Definition with Examples
- Molecular Orbital Theory - Formation of Molecular Orbitals, Linear Combination of Atomic Orbitals (LCAO)-Conditions for the Combination of Atomic Orbitals - Energy Level Diagrams for Molecular Orbitals -Bonding in Some Homonuclear Diatomic Molecules- H2, He2, Li2, B2, C2, N2 and O2
- Hydrogen Bonding-Cause of Formation of Hydrogen Bond - Types of Hydrogen Bonds Inter and Intramolecular-General Properties of Hydrogen Bonds
States Of Matter: Gases And Liquids
- Intermolecular Forces
- Thermal Energy
- Intermolecular Forces Vs Thermal Interactions
- The Gaseous State The Gas Laws
- Ideal Gas Equation
- Graham’s Law of Diffusion - Dalton’s Law of Partial Pressures
- Kinetic Molecular Theory of Gases
- Kinetic Gas Equation of an Ideal Gas (No derivation) Deduction of Gas Laws from Kinetic Gas Equation
- Distribution of Molecular Speeds, Average and Most Probable Speeds-Kinetic Energy of Gas Molecules.
- The Behaviour of Real Gases - Deviation from Ideal Gas Behaviour - Compressibility Factor Vs Pressure Diagrams of Real Gases.
- Liquefaction of Gases.
- Liquid State - Properties of Liquids in Terms of Intermolecular Interactions - Vapour Pressure, Viscosity, and Surface Tension (Qualitative Idea only. No Mathematical Derivation).
Stoichiometry
- Some Basic Concepts - Properties of Matter - Uncertainty in Measurement-Significant Figures, Dimensional Analysis
- Laws of Chemical Combinations - Law of Conservation of Mass, Law of Definite Proportions, Law of Multiple Proportions, Gay Lussac’s Law of Gaseous Volumes, Dalton’s Atomic Theory, Avogadro Law, Examples
- Atomic and Molecular Masses- Mole Concept and Molar Mass. Concept of Equivalent Weight
- Percentage Composition of Compounds and Calculations of Empirical and Molecular Formulae of Compounds Stoichiometry and Stoichiometric Calculations Limiting Reagent
- Methods of Expressing Concentrations of Solutions-Mass per-cent, Mole Fraction, Molarity, Molality, and Normality
- Redox Reactions-Classical Idea of Redox Reactions, Oxidation and Reduction Reactions-Redox Reactions in Terms of Electron Transfer
- Oxidation Number Concept
- Types of Redox Reactions-Combination, Decomposition, Displacement and Disproportionation Reactions Balancing of Redox Reactions - Oxidation Number Method Half Reaction (ion-electron) Method
- Redox Reactions in Titrimetry
Thermodynamics
- Thermodynamic Terms
- The System and the Surroundings
- Types of Systems and Surroundings
- The State of the System
- Internal Energy as a State Function. (a) Work (b) Heat (c) The General Case, the First Law of Thermodynamics Applications
- Work
- Enthalpy, H- a useful New State Function
- Extensive and Intensive Properties Heat Capacity
- The Relationship Between Cp and Cv
- Measurement of DU and DH: Calorimetry Enthalpy Change, Dr H of Reactions - Reaction Enthalpy (a) Standard Enthalpy of Reactions, (b) Enthalpy Changes during Transformations, (c) Standard Enthalpy of Formation, (d) Thermochemical equations (e) Hess’s law of constant heat summation Enthalpies for different types of reactions.
- (a) Standard enthalpy of combustion (∆c H q ), (b) Enthalpy of atomization (∆a H q ), phase transition, sublimation, and ionization, (c) Bond Enthalpy (∆bondH q ), (d) Enthalpy of solution (∆solH q ) and dilution-lattice enthalpy Spontaneity.
- (a) Is a decrease in enthalpy a criterion for spontaneity? (b) Entropy and spontaneity, the second law of thermodynamics, (c) Gibbs Energy and spontaneity
- Gibbs Energy change and equilibrium Absolute entropy and the third law of thermodynamics.
Chemical Equilibrium And Acids-Bases
- Equilibrium in Physical process
- Equilibrium in the chemical process - Dynamic Equilibrium
- Law of chemical Equilibrium - Law of mass action and Equilibrium constant
- Homogeneous Equilibria, Equilibrium constant in gaseous systems.
- Relationship between KP and Kc
- Heterogeneous Equilibria
- Applications of Equilibrium constant
- Relationship between Equilibrium constant K, reaction quotient Q and Gibbs energy G
- Factors affecting Equilibria. Principle application to the industrial synthesis of Ammonia and Sulphur trioxide
- Ionic Equilibrium in solutions
- Acids, bases and salts- Arrhenius, Bronsted-Lowry and Lewis concepts of acids and bases
- The ionization of Acids and Bases -Ionisation constant of water and its ionic product- pH scale-ionization constants of weak acid ionization of weak bases-relation between Ka and Kb -Di and polybasic acids and di and poly acidic Bases-Factors affecting acid strength-Common ion effect in the ionization of acids and bases Hydrolysis of salts and pH of their solutions
- Buffer solutions-designing of buffer solution-Preparation of Acidic buffer
- Solubility Equilibria of sparingly soluble salts
- Solubility product constant Common ion effect on the solubility of Ionic Salts
Hydrogen And Its Compounds
- Position of hydrogen in the periodic table
- Dihydrogen-Occurrence and Isotopes
- Preparation of Dihydrogen
- Properties of Dihydrogen
- Hydrides: Ionic, covalent, and non-stoichiometric hydrides
- Water: Physical properties
- Structure of water and ice.
- Chemical properties of water
- hard and soft water, Temporary and permanent hardness of water
- Hydrogen peroxide: Preparation
- Physical properties
- structure and chemical properties
- storage and uses
- Heavy Water
- Hydrogen as a fuel.
The S - Block Elements (Alkali And Alkaline Earth Metals)
Group 1 Elements
- Alkali metals
- Electronic configurations
- Atomic and Ionic Radii
- Ionization enthalpy
- Hydration enthalpy
- Physical properties
- Chemical properties
- Uses
- General characteristics of the compounds of the alkali metals: Oxides
- Halides
- Salts of oxo Acids
- Anomalous properties of Lithium: Differences and similarities with other alkali metals, Diagonal relationship
- Similarities between Lithium and Magnesium
- Some important compounds of Sodium: Sodium Carbonate Sodium Chloride
- Sodium Hydroxide
- Sodium hydrogen carbonate
- The biological importance of Sodium and Potassium.
Group 2 Elements
- Alkaline earth elements
- Electronic configuration
- Ionization enthalpy
- Hydration enthalpy
- Physical properties, Chemical properties
- Uses
- General characteristics of compounds of the Alkaline Earth Metals: Oxides, hydroxides, halides, salts of oxoacids (Carbonates Sulphates and Nitrates)
- Anomalous behaviour of Beryllium
- its diagonal relationship with Aluminium
- Some important compounds of calcium: Preparation and uses of Calcium Oxide Calcium Hydroxide
- Calcium Carbonate
- Plaster of Paris
- Cement
- The biological importance of Calcium and Magnesium.
P- Block Elements Group 13 (Boron Family)
- General introduction - Electronic configuration, Atomic radii, Ionization enthalpy, Electronegativity
- Physical & Chemical properties
- Important trends and anomalous properties of boron
- Some important compounds of boron - Borax, Orthoboric acid, diborane
- Uses of boron, aluminium, and their compounds.
p-Block Elements - Group 14 (Carbon Family)
- General introduction - Electronic configuration, Atomic radii, Ionization enthalpy, Electronegativity
- Physical & Chemical properties
- Important trends and anomalous properties of carbon
- Allotropes of carbon
- Uses of carbon
- Some important compounds of carbon and silicon -carbon-monoxide, carbon dioxide, Silica, silicones, silicates, and zeolites.
Environmental Chemistry
- Definition of terms: Air, Water, and Soil Pollutions
- Environmental Pollution
- Atmospheric pollution
- Tropospheric Pollution
- Gaseous Air Pollutants (Oxides of Sulphur Oxides of Nitrogen
- Hydrocarbons
- Oxides of Carbon (CO, CO2 ).
- Global warming and Greenhouse effect
- Acid Rain- Particulate Pollutants- Smog
- Stratospheric Pollution: Formation and breakdown of Ozone- Ozone hole- effects of depletion of the Ozone Layer
- Water Pollution: Causes of Water Pollution
- International standards for drinking water
- Soil Pollution: Pesticides, Industrial Wastes
- Strategies to control environmental pollution- waste Management- collection and disposal
- Green Chemistry: Green chemistry in day-to-day life
- Dry cleaning of clothes
- Bleaching of paper
- Synthesis of chemicals.
Organic Chemistry-Some Basic Principles And Techniques And Hydrocarbons
- General introduction
- Tetravalency of Carbon: shapes of organic compounds
- Structural representations of organic compounds
- Classification of organic compounds
- Nomenclature of organic compounds
- Isomerism
- Fundamental concepts in organic reaction mechanisms
- Fission of covalent bond
- Nucleophiles and electrophiles
- Electron movements in organic reactions
- Electron displacement effects in covalent bonds: inductive effect, resonance, resonance effect, electromeric effect, hyperconjugation
- Types of Organic reactions
- Methods of purification of organic compounds
- Qualitative elemental analysis of organic compounds
- Quantitative elemental analysis of organic compounds.
Hydrocarbons
- Classification of Hydrocarbons
- Alkanes - Nomenclature, isomerism (structural and conformations of ethane only)
- Preparation of alkanes
- Properties - Physical properties and chemical Reactivity, Substitution reactions - Halogenation(a free radical mechanism), Combustion, Controlled Oxidation, Isomerisation, Aromatization, reaction with steam and Pyrolysis
- Alkenes- Nomenclature, the structure of ethene, Isomerism (structural and geometrical)
- Methods of preparation
- Properties- Physical and chemical reactions: Addition of Hydrogen, halogen, water, sulphuric acid, Hydrogen halides (Mechanism- ionic and peroxide effect, Markovnikov’s, antiMarkovnikov’s or Kharasch effect).
- Oxidation, Ozonolysis, and Polymerization
- Alkynes - Nomenclature and isomerism, the structure of acetylene.
- Methods of preparation of acetylene
- Physical properties, Chemical reactions- the acidic character of acetylene, addition reactions- of hydrogen, Halogen, Hydrogen halides and water.
- Polymerization
- Aromatic Hydrocarbons:
- Nomenclature and isomerism, Structure of benzene, Resonance, and aromaticity
- Preparation of benzene.
- Physical properties.
- Chemical properties: Mechanism of electrophilic substitution.
- Electrophilic substitution reactions- Nitration, Sulphonation, Halogenation, Friedel-Craft’ alkylation and acylation
- Directive influence of functional groups in monosubstituted. benzene, Carcinogenicity, and toxicity.
Solid State
- General characteristics of solid-state Amorphous and crystalline solids
- Classification of crystalline solids based on different binding forces (molecular, ionic, metallic and covalent solids)
- Probing the structure of solids: X-ray crystallography
- Crystal lattices and unit cells.
- Bravais lattices primitive and centred unit cells.
- The number of atoms in a unit cell (primitive, body-centred and face-centred cubic unit cell).
- Close-packed structures: Close packing in one dimension, in two dimensions and three dimensions- tetrahedral and octahedral voids- the formula of a compound and number of voids filled- locating tetrahedral and octahedral voids.
- Calculations involving unit cell dimensions-density of the unit cell
- Imperfections in solids-types of point defects-stoichiometric and non-stoichiometric defects
- Electrical properties-conduction of electricity in metals, semiconductors, and insulators- band theory of metals
- Magnetic properties.
Solutions
- Types of solutions
- Expressing concentration of solutions - mass percentage, volume percentage, mass by volume percentage, parts per million, mole fraction, molarity and molality
- Solubility: Solubility of a solid in a liquid, solubility of a gas in a liquid, Henry’s law
- Vapour pressure of liquid solutions: vapour pressure of liquid-liquid solutions.
- Raoult’s law as a special case of Henry’s law - vapour pressure of solutions of solids in liquids
- Ideal and non-ideal solutions
- Colligative properties and determination of molar mass-relative lowering of vapour pressure-elevation of boiling point-depression of freezing point-osmosis and osmotic pressure-reverse osmosis and water purification
- Abnormal molar masses-van Hoff factor.
Electrochemistry And Chemical Kinetics Electrochemistry
- Electrochemical cells
- Galvanic cells: measurement of electrode potentials
- Nernst equation-equilibrium constant from Nernst equation- electrochemical cell and Gibbs energy of the cell reaction
- The conductance of electrolytic solutions- a measurement of the conductivity of ionic solutions-variation of conductivity and molar conductivity with concentration strong electrolytes and weak electrolytes-applications of Kohlrausch’s law
- Electrolytic cells and electrolysis: Faraday’s laws of electrolysis-products of electrolysis
- Batteries: primary batteries and secondary batteries
- Fuel cells
- Corrosion of metals-Hydrogen economy.
Chemical Kinetics
- Rate of a chemical reaction
- Factors influencing the rate of a reaction: dependence of rate on concentration- rate expression and rate constant- order of a reaction, molecularity of a reaction
- Integrated rate equations-zero order reactions-first order reactions-half life of a reaction
- Pseudo-first-order reaction
- Temperature dependence of the rate of a reaction -effect of catalyst
- Collision theory of chemical reaction rates.
Surface Chemistry
- Adsorption: Distinction between adsorption and absorption-mechanism of adsorption-types of adsorption-characteristics of physisorption-characteristics of chemisorptions-adsorption isotherms adsorption from solution phase-applications of adsorption
- Catalysis: Catalysts, promoters, and poisons-auto catalysis- homogeneous and heterogeneous catalysis-adsorption theory of heterogeneous catalysis-important features of solid catalysts: (a)activity (b)selectivity-shape-selective catalysis by zeolites-enzyme catalysis-characteristics and mechanism- catalysts in industry
- Colloids
- Classification of colloids: Classification based on the physical state of the dispersed phase and dispersion medium classification based on the nature of the interaction between the dispersed phase and dispersion medium classification based on the type of particles of the dispersed phase- multi molecular, macromolecular, and associated colloids- cleansing action of soaps-preparation of colloids-purification of colloidal solutions- properties of colloidal solutions: Colligative properties, Tyndal effect, colour, Brownian movement-charge on colloidal particles, electrophoresis
- Coagulation-precipitation methods coagulation of lyophilic sols and protection of colloids-Emulsions
- Colloids around us- application of colloids.
General Principles Of Metallurgy
- Occurrence of metals
- The concentration of ores-levigation, magnetic separation, froth flotation, leaching
- Extraction of crude metal from concentrated ore-conversion to oxide, reduction of oxide to the metal
- Thermodynamic principles of metallurgy – Ellingham diagram-limitations-application extraction of iron, copper, and zinc from their oxides
- Electrochemical principles of metallurgy
- Oxidation and reduction
- Refining of crude metal-distillation, liquation polling, electrolytic refining, zone refining and vapour phase refining
- Uses of aluminium, copper, zinc, and iron
P-Block Elements
Group-15 Elements
- Occurrence- electronic configuration, atomic and ionic radii, ionization enthalpy, electronegativity, physical and chemical properties
- Dinitrogen-preparation, properties, and uses
- Compounds of nitrogen-preparation, properties and uses of ammonia
- Oxides of nitrogen
- Preparation and properties of nitric acid
- Phosphorous-allotropic forms Phosphine-preparation, properties and uses
- Phosphorus halides
- Oxoacids of phosphorous
Group-16 Elements
- Occurrence- electronic configuration, atomic and ionic radii, ionization enthalpy, electron gain enthalpy, electronegativity, physical and chemical properties
- Dioxygenpreparation, properties, and uses
- Simple oxides
- Ozone-preparation, properties, structure, and uses
- Sulfur-allotropic forms
- Sulfur dioxide-preparation, properties and uses
- Oxoacids of Sulphur
- Sulphuric acid- manufacture, properties, and uses.
Group-17 Elements
- Occurrence, electronic configuration, atomic and ionic radii, ionization enthalpy, electron gain enthalpy, electronegativity, physical and chemical properties
- Chlorinepreparation, properties, and uses
- Hydrogen chloride- preparation, properties, and uses
- Oxoacids of halogens
- Interhalogen compounds- preparation, properties, and uses.
Group-18 Elements
- Occurrence, electronic configuration, ionization enthalpy, atomic radii, electron gain enthalpy, physical and chemical properties (a) Xenon-fluorine compoundsXeF2 ,XeF4 and XeF6 -preparation, hydrolysis and formation of fluoro anions-structures of XeF2 , XeF4 and XeF6 (b) Xenon-oxygen compounds XeO3 and XeOF4 - their formation and structures-uses of noble gases.
D And F Block Elements & Coordination Compounds
d and f Block Elements
- Position in the periodic table
- Electronic configuration of the d-block elements
- General properties of the transition elements (d-block) -physical properties, variation in atomic and ionic sizes of transition series, ionization enthalpies, oxidation states, trends in the M²+/M and M³+/M²+ standard electrode potentials, trends instability of higher oxidation states, chemical reactivity, and Eθ values, magnetic properties, the formation of coloured ions, formation of complex compounds, catalytic properties, the formation of interstitial compounds, alloy formation Some important compounds of transition elements-oxides and oxoanions of metals-preparation, properties and uses of potassium dichromate and potassium permanganate-structures of chromate, dichromate, manganate and permanganate ions
- Inner transition elements(f-block)-lanthanoids- electronic configuration-atomic and ionic sizes-oxidation states-general characteristics
- Actinoidselectronic configuration atomic and ionic sizes, oxidation states, general characteristics, and comparison with lanthanoids
- Some applications of d and f block elements.
Coordination Compounds
- Werner’s theory of coordination compounds
- Definitions of some terms used in coordination compounds
- Nomenclature of coordination compounds-IUPAC nomenclature
- Isomerism in coordination compounds- (a)Stereo isomerism-Geometrical and optical isomerism (b)Structural isomerism-linkage, coordination, ionization, and hydrate isomerism
- Bonding in coordination compounds. (a)Valence bond theory - magnetic properties of coordination compounds-limitations of valence bond theory (b) Crystal field theory (i) Crystal field splitting in octahedral and tetrahedral coordination entities (ii) Colour in coordination compounds-limitations of crystal field theory
- Bonding in metal carbonyls
- Stability of coordination compounds
- Importance and applications of coordination compounds.
Polymers
- Classification of Polymers -Classification based on source, structure, mode of polymerization, molecular forces and growth polymerization
- Types of polymerization reactions-addition polymerization or chain growth polymerization-ionic polymerization, free radical mechanic preparation of addition polymers-polythene, Teflon and polyacrylonitrile-condensation polymerization or step-growth polymerization-polyamides-preparation of Nylon 6,6 and nylon 6-poly esters-terylene-bakelite, melamine-formaldehyde polymers
- copolymerization-Rubber-natural rubber-vulcanization of rubber-Synthetic rubbers-preparation of neoprene and Buna-N
- The molecular mass of polymers-number average and weight average molecular masses- polydispersity index(PDI)
- Biodegradable polymers-PHBV, Nylon 2-nylon 6
- Polymers of commercial importance polypropene, polystyrene, polyvinylchloride (PVC), urea-formaldehyde resin, glyptal, and Bakelite - their monomers, structures, and uses.
Biomolecules
- Carbohydrates - Classification of Carbohydrates- Monosaccharides: preparation of glucose from sucrose and starch- Properties and structure of glucose- D, L configurations and (+), (-) notations of glucose-Structure of fructose
- Disaccharides: Sucrose- preparation, structure
- Invert sugar- Structures of maltose and lactose-Polysaccharides: Structures of starch, cellulose, and glycogen- Importance of carbohydrates
- Proteins-Aminoacids: Natural aminoacids-classification of amino acids - structures and D and L forms- Zwitter ions
- Proteins: Structures, classification, fibrous and globular- primary, secondary, tertiary and quaternary structures of proteins- Denaturation of proteins
- Enzymes: Enzymes, mechanism of enzyme action
- Vitamins: Explanation-names classification of vitamins - sources of vitamin-deficiency diseases of different types of vitamins
- Nucleic acids: chemical composition of nucleic acids, structures of nucleic acids, DNA fingerprinting biological functions of nucleic acids Hormones: Definition, different types of hormones, their production, biological activity, diseases due to their abnormal activities.
Chemistry In Everyday Life
- Drugs and their classification: (a) Classification of drugs based on pharmacological effect (b) Classification of drugs based on drug action (c) Classification of drugs based on chemical structure (d) Classification of drugs based on molecular targets
- Drug-Target interaction enzymes as drug targets (a) Catalytic action of enzymes (b) Drug-enzyme interaction, receptors as drug targets
- The therapeutic action of different classes of drugs: antacids, antihistamines, neurologically active drugs: tranquillizers, analgesics-non-narcotic, narcotic analgesics, antimicrobials-antibiotics, antiseptics, and disinfectants- antifertility drugs
- Chemicals in food-artificial sweetening agents, food preservatives, antioxidants in food
- Cleansing agents-soaps and synthetic detergents – types and examples.
Haloalkanes And Haloarenes
- Classification and nomenclature
- Nature of C-X bond
- Methods of preparation: Alkyl halides and aryl halides-from alcohols, from hydrocarbons (a) by free radical halogenation (b) by electrophilic substitution (c) by replacement of diazonium group(Sandmeyer reaction) (d) by the addition of hydrogen halides and halogens to alkenes-by halogen exchange reactions Physical properties melting and boiling points, density and solubility
- Chemical reactions: Reactions of haloalkanes (i)Nucleophilic substitution reactions (a) SN² mechanism (b) SN¹ mechanism (c) stereochemical aspects of nucleophilic substitution reactions-optical activity (ii) Elimination reactions (iii) Reaction with metals-Reactions of haloarenes: (i) Nucleophilic substitution (ii)Electrophilic substitution and (iii) Reaction with metals
- Polyhalogen compounds: Uses and environmental effects of dichloromethane, trichloromethane triiodomethane, tetrachloromethane, freons, and DDT.
Organic Compounds Containing C, H, And O
Alcohols, Phenols, And Ethers
- Alcohols, phenols, and ethers -classification
- Nomenclature: (a)Alcohols, (b)phenols and (c) ethers Structures of hydroxy and ether functional groups
- Methods of preparation: Alcohols from alkenes and carbonyl compounds, from Grignard reagents
- Phenols from haloarenes, benzene sulphonic acid, diazonium salts, cumene
- Physical properties of alcohols and phenols
- Chemical reactions of alcohols and phenols (i) Reactions involving cleavage of O-H bond in alcohols-Acidity of alcohols and phenols, esterification (ii) Reactions involving cleavage of C-O bond- reactions with HX, PX3, dehydration and oxidation (iii) Reactions of phenols- electrophilic aromatic substitution, Kolbe’s reaction, Reimer - Tiemann reaction, reaction with zinc dust, oxidation
- Commercially important alcohols (methanol, ethanol)
- Ethers-Methods of preparation: By dehydration of alcohols, Williamson synthesis- Physical properties-Chemical reactions: Cleavage of C-O bond and electrophilic substitution of aromatic ethers (anisole).
Aldehydes And Ketones
- Nomenclature and structure of carbonyl group
- Preparation of aldehydes and ketones-(1) by oxidation of alcohols (2) by dehydrogenation of alcohols (3) from hydrocarbons -Preparation of aldehydes (1) from acyl chlorides (2) from nitriles and esters(3) from hydrocarbons-Preparation of ketones(1) from acyl chlorides (2)from nitriles (3)from benzene or substituted benzenes
- Physical properties of aldehydes and ketones
- Chemical reactions of aldehydes and ketones-nucleophilic addition, reduction, oxidation, reactions due to α - Hydrogen and other reactions (Cannizzaro reaction, electrophilic substitution reaction)
- Uses of aldehydes and ketones.
Carboxylic Acids
- Nomenclature and structure of carboxyl group
- Methods of preparation of carboxylic acids (1)from primary alcohols and aldehydes (2) from alkylbenzenes (3)from nitriles and amides (4)from Grignard reagents (5) from acyl halides and anhydrides (6) from esters
- Physical properties
- Chemical reactions: (i) Reactions involving cleavage of O-H bond-acidity, reactions with metals and alkalies (ii) Reactions involving cleavage of C-OH bond-formation of anhydride, reactions with PCl5, PCl3, SOCl2, esterification and reaction with ammonia (iii) Reactions involving-COOH group-reduction, decarboxylation (iv) Substitution reactions in the hydrocarbon part - halogenation and ring substitution
- Uses of carboxylic acids.
Organic Compounds Containing Nitrogen
Amines
- Structure of amines
- Classification
- Nomenclature
- Preparation of amines: reduction of nitro compounds, ammonolysis of alkyl halides, reduction of nitriles, reduction of amides, Gabriel phthalimide synthesis, and Hoffmann bromamide degradation reaction
- Physical properties
- Chemical reactions: the basic character of amines, alkylation, acylation, carbonyl amine reaction, reaction with nitrous acid, reaction with aryl sulphonyl chloride, the electrophilic substitution of aromatic amines (aniline)-bromination, nitration, and sulphonation.
Diazonium Salts
- Methods of preparation of diazonium salts (by diazotization) Physical properties
- Chemical reactions: Reactions involving the displacement of Nitrogen
- Sandmeyer reaction, Gatterman reaction, replacement by i) iodide and fluoride ions ii) hydrogen, hydroxyl, and Nitro groups reactions involving retention of the diazo group
- Coupling reactions
- The importance of diazonium salts in the synthesis of aromatic compounds.
Cyanides And Isocyanides
- Structure and nomenclature of cyanides and isocyanides Preparation, physical properties and chemical reactions of cyanides and isocyanides.
AP EAMCET 2025 Physics Syllabus
The physics syllabus for 2025 AP EAMCET has been mentioned below of all the topics.
Units And Measurements
- The international system of units
- Measurement of Length
- Measurement of Large Distances
- Estimation of Very Small Distances
- Size of a Molecule
- Range of Lengths
- Measurement of Mass
- Range of Masses
- Measurement of time
- Accuracy
- The precision of instruments and errors in measurement
- Systematic errors
- Random errors
- Least count error
- Absolute Error
- Relative Error and Percentage Error
- Combination of Errors
- Significant figures
- Rules for Arithmetic Operations with Significant Figures
- Rounding off the Uncertain Digits
- Rules for Determining the Uncertainty in the Results of Arithmetic Calculations
- Dimensions of Physical Quantities
- Dimensional Formulae and dimensional equations
- Dimensional Analysis and its Applications
- Checking the Dimensional Consistency of Equations
- Deducing Relation among the Physical Quantities
Motion In A Straight Line
- Position
- path length and displacement
- Average velocity and average speed
- Instantaneous velocity and speed
- Acceleration, Kinematic equations for uniformly accelerated motion
- Relative velocity
Motion In A Plane
- Scalars and vectors
- Position and displacement vectors
- Equality of vectors
- Multiplication of vectors by real numbers
- Addition and subtraction of vectors - graphical method
- Resolution of vectors
- Vector addition - analytical method
- Motion in a plane
- Position vector and displacement
- Velocity, acceleration
- Motion in a plane with constant acceleration
- Relative velocity in two dimensions
- Projectile motion
- Equation of path of a projectile
- Time of maximum height
- Maximum height of a projectile
- Horizontal range of a projectile
- Uniform circular motion
Laws Of Motion
- Aristotle’s fallacy
- The law of inertia
- Newton’s first law of motion
- Newton’s second law of motion, momentum
- Impulse
- Newton’s third law of motion
- Conservation of momentum
- Equilibrium of a particle
- Common forces in mechanics
- Friction, types of friction
- Static, kinetic, and rolling frictions
- Circular motion
- The motion of a car on a level road
- The motion of a car on a banked road, solving problems in mechanics
Work, Energy, And Power
- The Scalar Product
- Notions of work and kinetic energy
- The work-energy theorem
- Work, Kinetic energy
- Work is done by a variable force
- The work-energy theorem for a variable force
- The concept of Potential Energy
- The conservation of Mechanical Energy
- The Potential Energy of a spring
- Various forms of energy
- Heat
- Chemical Energy
- Electrical Energy
- The Equivalence of Mass and Energy
- Nuclear Energy
- The Principle of Conservation of Energy
- Power, Collisions
- Elastic and Inelastic Collisions
- Collisions in one dimension
- Coefficient of Restitution and its determination
- Collisions in Two Dimensions
Systems Of Particles And Rotational Motion
- Rigid body motion
- Centre of mass
- Centre of Gravity
- The motion of the Centre of mass
- Linear momentum of a system of particles
- Vector product of two vectors
- Angular velocity and its relation with linear velocity
- Angular acceleration
- Kinematics of rotational motion about a fixed axis
- Moment of force (Torque)
- Angular momentum of a particle
- Torque and angular momentum for a system of particles
- Conservation of angular momentum
- Equilibrium of a rigid body
- Principle of moments
- Moment of inertia
- Theorems of perpendicular and parallel axes
- Dynamics of rotational motion about a fixed axis
- Angular momentum in case of rotation about a fixed axis
- Rolling motion
- Kinetic Energy of Rolling Motion
Oscillations
- Periodic and oscillatory motions
- Period and frequency
- Displacement
- Simple harmonic motion (S.H.M.)
- Simple harmonic motion and uniform circular motion
- Velocity and acceleration in simple harmonic motion
- Force law for Simple Harmonic Motion
- Energy in simple harmonic motion
- Some systems executing Simple Harmonic Motion
- Oscillations due to a spring
- The Simple Pendulum
- Damped simple harmonic motion
- Forced oscillations and resonance
Gravitation
- Kepler’s laws
- Universal law of gravitation
- central forces
- the gravitational constant
- Acceleration due to gravity of the earth
- Acceleration due to gravity below and above the surface of earth
- Gravitational potential energy
- Escape speed
- Orbital Speed
- Earth satellites
- Energy of an orbiting satellite
- Geostationary and polar satellites
- Weightlessness
Mechanical Properties Of Solids
- Elastic behaviour of solids
- Stress and strain
- Hooke’s law
- Stress-strain curve
- Elastic Moduli
- Young’s Modulus
- Determination of Young’s Modulus of the Material of a Wire
- Shear Modulus
- Bulk Modulus
- Poisson’s Ratio
- Elastic Potential Energy in a Stretched wire
- Applications of elastic behaviour of materials
Mechanical Properties Of Fluids
- Pressure
- Pascal’s Law
- Variation of Pressure with Depth
- Atmospheric Pressure and Gauge Pressure
- Hydraulic Machines
- Archimedes’ Principle
- Streamline flow
- Bernoulli’s principle
- Speed of Efflux
- Torricelli’s Law
- Venturi-meter
- Blood Flow and Heart Attack
- Dynamic Lift
- Viscosity
- Variation of Viscosity of fluids with temperature
- Stokes’ Law
- Reynolds number
- Critical Velocity
- Surface tension and Surface Energy
- Angle of Contact
- Drops and Bubbles
- Capillary Rise
- Detergents and Surface Tension
Thermal Properties Of Matter
- Temperature and heat
- Measurement of temperature
- Ideal-gas equation and absolute temperature
- Thermal expansion
- Specific heat capacity
- Calorimetry
- Change of state
- Triple Point
- Regulation
- Latent Heat
- Heat transfer
- Conduction
- Convection
- Radiation
- Black body Radiation
- Greenhouse Effect
- Newton’s law of cooling and its experimental verification
Thermodynamics
- Thermal equilibrium
- Zeroth law of thermodynamics
- Heat
- Internal Energy and work
- First law of thermodynamics
- Specific heat capacity
- Specific heat capacity of water
- Thermodynamic state variables and Equation of State
- Thermodynamic processes
- Quasi-static process
- Isothermal Process
- Adiabatic Process
- Isochoric Process
- Isobaric process
- Cyclic process
- Heat engines
- Refrigerators and heat pumps
- Second law of thermodynamics
- Reversible and irreversible processes
- Carnot engine
- Carnot’s theorem
Kinetic Theory
- Molecular nature of matter
- Behaviour of gases
- Boyle’s Law, Charles’ Law
- Kinetic theory of an ideal gas
- Pressure of an Ideal Gas
- Kinetic interpretation of temperature
- Law of equipartition of energy
- Specific heat capacity
- Monatomic Gases
- Diatomic Gases
- Polyatomic Gases
- Specific Heat Capacity of Solids
- Specific Heat Capacity of Water
- Mean free path
Waves
- Transverse and longitudinal waves
- Displacement Relation In A Progressive Wave
- Amplitude And Phase
- Wavelength And Angular Wave Number
- Period
- Angular Frequency And Frequency
- The Speed Of A Travelling Wave
- Speed Of A Transverse Wave On Stretched String
- Speed Of A Longitudinal Wave (Speed Of Sound)
- The Principle Of Superposition Of Waves
- Reflection Of Waves
- Standing Waves And Normal Modes
- Beats
- Doppler Effect: Source Moving And Observer Stationary
- Observer Moving And Source Stationary
- Both Source And Observer Moving
Ray Optics And Optical Instruments
- Reflection Of Light By Spherical Mirrors
- Sign Convention
- Focal Length Of Spherical Mirrors
- The Mirror Equation
- Refraction
- Total Internal Reflection
- Total Internal Reflection In Nature And Its Technological Applications
- Refraction At Spherical Surfaces And By Lenses, Power Of A Lens
- Combination Of Thin Lenses In Contact, Refraction Through A Prism
- Dispersion By A Prism
- Some Natural Phenomena Due To Sunlight
- The Rainbow, Scattering Of Light
- Optical Instruments
- The Eye
- The Simple And Compound Microscopes
- Refracting Telescope And Cassegrain Reflecting Telescope
Wave Optics
- Huygens Principle
- Refraction And Reflection Of Plane Waves Using Huygens Principle
- Refraction In A Rarer Medium (At The Denser Medium Boundary)
- Reflection Of A Plane Wave By A Plane Surface
- The Doppler Effect
- Coherent And Incoherent Addition Of Waves
- Interference Of Light Waves And Young‘S Experiment, Diffraction
- The Single Slit Diffraction
- Resolving Power Of Optical Instruments
- The Validity Of Ray Optics
- Polarisation
- Polarisation By Scattering
- Polarisation By Reflection
Electric Charges And Fields
- Electric Charge
- Conductors And Insulators
- Charging By Induction
- Basic Properties Of Electric Charges
- Additivity Of Charges
- Conservation Of Charge
- Quantization Of Charge
- Coulomb’s Law
- Forces Between Multiple Charges
- Electric Field
- Electric Field Due To A System Of Charges
- Physical Significance Of Electric Field
- Electric Field Lines
- Electric Flux
- Electric Dipole
- The Field Of An Electric Dipole For Points On The Axial Line And On The Equatorial Plane
- Physical Significance Of Dipoles
- Dipole In A Uniform External Field
- Continuous Charge Distribution
- Gauss’s Law
- Applications Of Gauss’s Law
- Field Due To An Infinitely Long Straight Uniformly Charged Wire
- Field Due To A Uniformly Charged Infinite Plane Sheet
- Field Due To A Uniformly Charged Thin Spherical Shell
Electrostatic Potential And Capacitance
- Electrostatic Potential
- Potential Due To A Point Charge
- Potential Due To An Electric Dipole
- Potential Due To A System Of Charges
- Equipotential Surfaces
- Relation Between Field And Potential
- Potential Energy Of A System Of Charges
- Potential Energy In An External Field
- Potential Energy Of A Single Charge
- Potential Energy Of A System Of Two Charges In An External Field
- Potential Energy Of A Dipole In An External Field
- Electrostatics Of Conductors
- Electrostatic Shielding
- Dielectrics And Polarisation
- Electric Displacement
- Capacitors And Capacitance
- The Parallel Plate Capacitor
- Effect Of Dielectric On Capacitance
- Combination Of Capacitors
- Capacitors In Series
- Capacitors In Parallel
- Energy Stored In A Capacitor
- Van De Graaff Generator
Current Electricity
- Electric Current
- Electric Current In Conductors
- Ohm’s Law
- Drift Of Electrons And The Origin Of Resistivity
- Mobility
- Limitations Of Ohm’s Law
- Resistivity Of Various Materials
- Colour Code Of Resistors
- Temperature Dependence Of Resistivity
- Electrical Energy
- Power
- Combination Of Resistors – Series And Parallel
- Cells
- EMF
- Internal Resistance
- Cells In Series And In Parallel
- Kirchhoff’s Rules
- Wheatstone Bridge
- Meter Bridge
- Potentiometer
Moving Charges And Magnetism
- Magnetic Force
- Sources And Fields
- Magnetic Field
- Lorentz Force
- Magnetic Force On A Current Carrying Conductor
- Motion In A Magnetic Field
- Helical Motion Of Charged Particles
- Motion In Combined Electric And Magnetic Fields
- Velocity Selector
- Cyclotron
- Magnetic Field Due To A Current Element
- Biot – Savart’s Law
- Magnetic Field On The Axis Of A Circular Current Loop
- Ampere’s Circuital Law
- The Solenoid And The Toroid
- Force Between Two Parallel Current-Carrying Conductors
- The Ampere (UNIT)
- Torque On Current Loop
- Magnetic Dipole
- Torque On A Rectangular Current Loop In A Uniform Magnetic Field
- Circular Current Loop As A Magnetic Dipole
- The Magnetic Dipole Moment Of A Revolving Electron
- The Moving Coil Galvanometer
- Conversion Into Ammeter And Voltmeter
Magnetism And Matter
- The Bar Magnet
- The Magnetic Field Lines
- Bar Magnet As An Equivalent Solenoid
- The Dipole In A Uniform Magnetic Field
- The Electrostatic Analog
- Magnetism And Gauss’s Law
- The Earth’s Magnetism
- Magnetic Declination And Dip
- Magnetisation And Magnetic Intensity
- Susceptibility
- Magnetic Properties Of Materials
- Diamagnetism
- Paramagnetism
- Ferromagnetism
- Hysteresis Loop
- Permanent Magnets And Electromagnets
Electromagnetic Induction
- The Experiments Of Faraday And Henry
- Magnetic Flux
- Faraday’s Law Of Induction
- Lenz’s Law And Conservation Of Energy
- Motional Electromotive Force
- Energy Consideration - A Quantitative Study
- Eddy Currents
- Inductance
- Mutual Inductance
- Self Inductance
- AC Generator
Alternating Current
- AC Voltage Applied To A Resistor
- Representation Of AC Current And Voltage By Rotating Vectors - Phasors
- AC Voltage Applied To An Inductor
- AC Voltage Applied To A Capacitor
- AC Voltage Applied To A Series LCR Circuit
- Phasor – Diagram Solution
- Analytical Solution
- Resonance
- Sharpness Of Resonance
- Power In AC Circuit
- The Power Factor
- LC Oscillations
- Transformers
Electromagnetic Waves
- Displacement Current
- Maxwell’s Equations
- Electromagnetic Waves
- Sources Of Electromagnetic Waves
- Nature Of Electromagnetic Waves
- Electromagnetic Spectrum: Radio Waves
- Microwaves
- Infrared Waves
- Visible Rays
- Ultraviolet Rays
- X-Rays
- Gamma Rays
Dual Nature Of Radiation And Matter
- Electron Emission
- Photoelectric Effect
- Hertz’s Observations
- Hallwachs And Lenard’s Observations
- Experimental Study Of Photoelectric Effect
- Effect Of Intensity Of Light On Photocurrent
- Effect Of Potential On Photoelectric Current
- Effect Of Frequency Of Incident Radiation On Stopping Potential
- Photoelectric Effect And Wave Theory Of Light
- Einstein’s Photoelectric Equation
- Energy Quantum Of Radiation
- Particle Nature Of Light
- The Photon
- Wave Nature Of Matter
- Photocell
- Davisson And Germer Experiment
Atoms
- Alpha Particle Scattering And Rutherford’s Nuclear Model Of Atom
- Alpha Particle Trajectory
- Electron Orbits
- Atomic Spectra
- Spectral Series
- Bohr Model Of The Hydrogen Atom
- Energy Levels
- Franck – Hertz Experiment
- The Line Spectra Of The Hydrogen Atom
- De Broglie’s Explanation Of Bohr’s Second Postulate Of Quantization
- Laser Light
Nuclei
- Atomic Masses And Composition Of Nucleus
- Discovery Of Neutron
- Size Of The Nucleus
- Mass - Energy And Nuclear Binding Energy
- Nuclear Force
- Radioactivity
- Law Of Radioactive Decay
- Alpha Decay
- Beta Decay
- Gamma Decay
- Nuclear Energy
- Fission
- Nuclear Reactor
- Nuclear Fusion
- Energy Generation In Stars
- Controlled Thermonuclear Fusion
Semiconductor Electronics: Materials, Devices And Simple Circuits
- Classification Of Metals
- Conductors
- And Semiconductors On The Basis Of Conductivity And Energy Bands
- Band Theory Of Solids
- Intrinsic Semiconductor
- Extrinsic Semiconductor
- P-Type Semiconductor
- N-Type Semiconductor
- P-N Junction Formation
- Semiconductor Diode
- P-N Junction Diode Under Forward Bias
- P-N Junction Diode Under Reverse Bias
- Application Of Junction Diode As A Rectifier
- Special Purpose P-N Junction Diodes
- Zener Diode
- Zener Diode As Voltage Regulator
- Optoelectronic Junction Devices
- Photodiode
- Light Emitting Diode
- Solar Cell
- Junction Transistor
- Structure And Action
- Basic Transistor Circuit Configurations And Transistor Characteristics
- Transistor As A Switch And As An Amplifier (CE – Configuration)
- A Feedback Amplifier And Transistor Oscillator
- Digital Electronics And Logic Gates
- NOT, OR, AND, NAND And NOR Gates, Integrated Circuits
Communication Systems
- Elements Of A Communication System
- Basic Terminology Used In Electronic Communication Systems
- Bandwidth Of Signals
- Bandwidth Of Transmission Medium
- Propagation Of Electromagnetic Waves
- Ground Waves
- Sky Waves
- Space Wave
- Modulation And Its Necessity
- Size Of The Antenna Or Aerial
- Effective Power Radiated By An Antenna
- Mixing Up Of Signals From Different Transmitters
- Amplitude Modulation
- Production Of Amplitude Modulated Wave
- Detection Of Amplitude Modulated Wave
AP EAMCET 2025 Maths Syllabus
The math syllabus for the AP EAMCET 2025 has been listed below.
Algebra
- Functions: Types of functions – Definitions - Inverse functions and Theorems - Domain, Range, Inverse of real-valued functions.
- Mathematical Induction: Principle of Mathematical Induction & Theorems - Applications of Mathematical Induction - Problems on divisibility.
- Matrices: Types of matrices - Scalar multiple of a matrix and multiplication of matrices - Transpose of a matrix - Determinants - Adjoint and Inverse of a matrix - Consistency and inconsistency of Equations- Rank of a matrix - solution of simultaneous linear equations.
- Complex Numbers: Complex number as an ordered pair of real numbers- fundamental operations - Representation of complex numbers in the form a+ib - Modulus and amplitude of complex numbers –Illustrations - Geometrical and Polar Representation of complex numbers in Argand
- De Moivre’s Theorem: De Moivre’s theorem- Integral and Rational indices - nth roots of unit geometrical Interpretations – Illustrations.
- Quadratic Expressions: Quadratic expressions, equations in one variable - Sign of quadratic expressions – Change in signs – Maximum and minimum values - Quadratic inequations.
- Theory of Equations: The relation between the roots and coefficients in an equation - Solving the equations when two or more roots of it are connected by certain relation - Equation with real coefficients, the occurrence of complex roots in conjugate pairs and its consequences - Transformation of equations - Reciprocal Equations.
- Permutations and Combinations: Fundamental Principle of counting – linear and circular permutations- Permutations of ‘n’ dissimilar things taken ‘r’ at a time - Permutations when repetitions allowed - Circular permutations - Permutations with constraint repetitions - Combinations-definitions, certain theorems, and their applications.
- Binomial Theorem: Binomial theorem for positive integral index - Binomial theorem for rational Index (without proof) - Approximations using Binomial theorem.
- Partial fractions: Partial fractions of f(x)/g(x) when g(x) contains non repeated linear factors - Partial fractions of f(x)/g(x) where both f(x) and g(x) are polynomials and when g(x) contains repeated and/or non-repeated linear factors - Partial fractions of f(x)/g(x) when g(x) contains irreducible factors.
Trigonometry
- Trigonometric Ratios up to Transformations: Graphs and Periodicity of Trigonometric functions - Trigonometric ratios and Compound angles - Trigonometric ratios of multiple and sub-multiple angles - Transformations - Sum and Product rules.
- Trigonometric Equations: General Solution of Trigonometric Equations - Simple Trigonometric Equations – Solutions.
- Inverse Trigonometric Functions: To reduce a Trigonometric Function into a bijection - Graphs of Inverse Trigonometric Functions - Properties of Inverse Trigonometric Functions.
- Hyperbolic Functions: Definition of Hyperbolic Function – Graphs - Definition of Inverse Hyperbolic Functions – Graphs - Addition formulae of Hyperbolic Functions.
- Properties of Triangles: Relation between sides and angles of a Triangle - Sine, Cosine, Tangent and Projection rules - Half angle formulae and areas of a triangle – Incircle and Excircle of a Triangle.
Vector Algebra
- The addition of Vectors: Vectors as a triad of real numbers - Classification of vectors - Addition of vectors - Scalar multiplication - Angle between two non zero vectors - Linear combination of vectors - Component of a vector in three dimensions - Vector equations of line and plane including their Cartesian equivalent forms.
- Product of Vectors: Scalar Product - Geometrical Interpretations - orthogonal projections - Properties of dot product - Expression of the dot product In i, j, k system - Angle between two vectors - Geometrical Vector methods - Vector equations of the plane in normal form - Angle between two planes - Vector product of two vectors and properties - Vector product in i, j, k system - Vector Areas - Scalar Triple Product - Vector equations of the plane in different forms, skew lines, shortest distance and their Cartesian equivalents. The plane through the line of intersection of two planes, condition for coplanarity of two lines, perpendicular distance of a point from a plane, the angle between the line and a plane. Cartesian equivalents of all these results - Vector Triple Product – Results.
Measures Of Dispersion And Probability
- Measures of Dispersion - Range - Mean deviation - Variance and standard deviation of ungrouped/grouped data - Coefficient of variation and analysis of frequency distribution with equal means but different variances.
- Probability: Random experiments and events - Classical definition of probability, Axiomatic approach and addition theorem of probability - Independent and dependent events - conditional probability- multiplication theorem and Baye’s theorem and applications.
- Random Variables and Probability Distributions: Random Variables - Theoretical discrete distributions – Binomial and Poisson Distributions.
Coordinate Geometry
- Locus: Definition of locus – Illustrations - To find equations of locus - Problems connected to it.
- Transformation of Axes: Transformation of axes - Rules, Derivations, and Illustrations - Rotation of axes - Derivations – Illustrations.
- The Straight Line: Revision of fundamental results - Straight line - Normal form – Illustrations - Straight line - Symmetric form - Straight line - Reduction into various forms - Intersection of two Straight Lines - Family of straight lines - Concurrent lines - Condition for Concurrent lines - Angle between two lines - Length of the perpendicular from a point to a Line - Distance between two parallel lines - Concurrent lines - properties related to a triangle.
- Pair of Straight lines: Equations of pair of lines passing through origin - the angle between a pair of lines - Condition for perpendicular and coincident lines, bisectors of angles - Pair of bisectors of angles - Pair of lines - second-degree general equation - Conditions for parallel lines - the distance between them, Point of the intersection of pair of lines - Homogenizing a second-degree equation with a first-degree equation in x and y.
- Circle: Equation of circle -standard form-centre and radius equation of a circle with a given line segment as diameter & equation of a circle through three non-collinear points - parametric equations of a circle - Position of a point in the plane of a circle – the power of a point-definition of tangent-length of a tangent - Position of a straight line in the plane of a circle-conditions for a line to be tangent – chord joining two points on a circle – equation of the tangent at a point on the circle- point of contact equation of normal - Chord of contact - pole and polar-conjugate points and conjugate lines - equation of chord in term of its midpoint - Relative position of two circles- circles touching each other externally, internally- common tangents – centres of similitude- equation of pair of tangents from an external point.
- A system of circles: Angle between two intersecting circles - Radical axis of two circles properties- Common chord and common tangent of two circles – radical centre.
- Parabola: Conic sections –Parabola- the equation of the parabola in standard form different forms of the parabola- parametric equations - Equations of tangent and normal at a point on the parabola ( Cartesian and parametric) - conditions for a straight line to be a tangent.
- Ellipse: Equation of ellipse in standard form- Parametric equations - Equation of tangent and normal at a point on the ellipse (Cartesian and parametric) - condition for a straight line to be a tangent.
- Hyperbola: Equation of hyperbola in standard form- Parametric equations - Equations of tangent and normal at a point on the hyperbola (Cartesian and parametric) - conditions for a straight line to be a tangent- Asymptotes.
- Three Dimensional Coordinates: Coordinates - Section formulae - Centroid of a triangle and tetrahedron.
- Direction Cosines and Direction Ratios: Direction Cosines - Direction Ratios.
- Plane: Cartesian equation of Plane - Simple Illustrations.
Calculus
- Limits and Continuity: Intervals and neighbourhoods – Limits - Standard Limits – Continuity
- Differentiation: Derivative of a function - Elementary Properties - Trigonometric, Inverse Trigonometric, Hyperbolic, Inverse Hyperbolic Function – Derivatives - Methods of Differentiation - Second-Order Derivatives
- Applications of Derivatives: Errors and approximations - Geometrical Interpretation of a derivative - Equations of tangents and normals - Lengths of a tangent, normal, subtangent and subnormal - Angles between two curves and condition for orthogonality of curves - Derivative as Rate of change - Rolle’s Theorem and Lagrange’s Mean value theorem without proofs and their geometrical interpretation - Increasing and decreasing functions - Maxima and Minima
- Integration: Integration as the inverse process of differentiation- Standard forms -properties of integrals - Method of substitution- integration of Algebraic, exponential, logarithmic, trigonometric and inverse trigonometric functions - Integration by parts – Integration by Partial fractions method - Reduction formulae
- Definite Integrals: Definite Integral as the limit of the sum - Interpretation of Definite Integral as an area - Fundamental Theorem of Integral Calculus (without proof) – Properties - Reduction formulae - Application of Definite integral to areas
- Differential equations: Formation of differential equation-Degree and order of an ordinary differential equation - Solving differential equation by i) Variables separable method, ii) Homogeneous differential equation, iii) Non - Homogeneous differential equation, iv) Linear differential equations.
AP EAMCET 2025 Medical Syllabus (Biology)
The Biology section comprises of Botany and Zoology.
Botany Syllabus
The Botany syllabus for the 2025 AP EAMCET has been listed below.
- The living world
- Biological Classification
- Science of plants – Botany
- Plant Kingdom
Structural Organisation in Plants- Morphology
- Morphology of Flowering Plants
- Vegetative: Parts of a typical Angiospermic plant; Vegetative morphology and modifications- Root, Stem, and Leaf- types; Venation, Phyllotaxy.
- Reproductive: Inflorescence - Racemose, Cymose, and special types
- Flower
- Fruits
Reproduction in Plants
- Modes of reproduction
- Sexual Reproduction in Flowering Plants
Plant Systematics
- Taxonomy of angiosperms
Cell Structure and Function
- Cell-The unit if life
- Biomolecules
- Cell Cycle and Cell Division
Internal Organisation in Plants
- Histology and Anatomy of Flowering Plants
Plant Ecology
- Ecological Adaptations, Succession, and Ecological Services
Plant Physiology
- Transport in plants
- Plant water relations
- Long-distance transport of water
- Mineral nutrition
- Enzymes
- Photosynthesis in Higher Plants
- respiration of plants
- Plant growth and development
Micro-Biology
- Bacteria
- Viruses
Genetics
- Principles of genetics and variation
Molecular Biology
- Molecular basis of inheritance
Biotechnology
- Principles and processes of Biotechnology
- Biotechnology and its application
Plants, Microbes and Human Welfare
- Strategies for enhancement of food
- Microbes in Human welfare.
Zoology Syllabus
The zoology syllabus for the AP EAMCET 2025 has been listed below.
- Diversity of the living world
- Structural Organisation in Animals
- Animal diversity-Invertebrate Phyla
- Animal diversity-phylum Chordata
- Locomotion and reproduction in protozoa
- Biology and human welfare
- Type study of Periplaneta americana
- Ecology and environment
- Human anatomy and physiology-I
- Human anatomy and physiology-II
- Human anatomy and physiology-III
- Human anatomy and physiology-IV
- Human reproduction
- Reproductive health
- Human reproduction system
- Genetics
- Organic evolution
- Applied biology
Also Read: AP EAMCET Preparation Tips
AP EAMCET 2025 Topic-Wise Weightage
AP EAMCET syllabus 2025 topic weightage has been shared below for different EAMCET subjects for the candidates. The topic-wise weightage list will help candidates learning the importance of each topic and will guide them with a correct pattern of preparation as well. Candidates must check the AP EAMCET chapter-wise weightage details shared below:
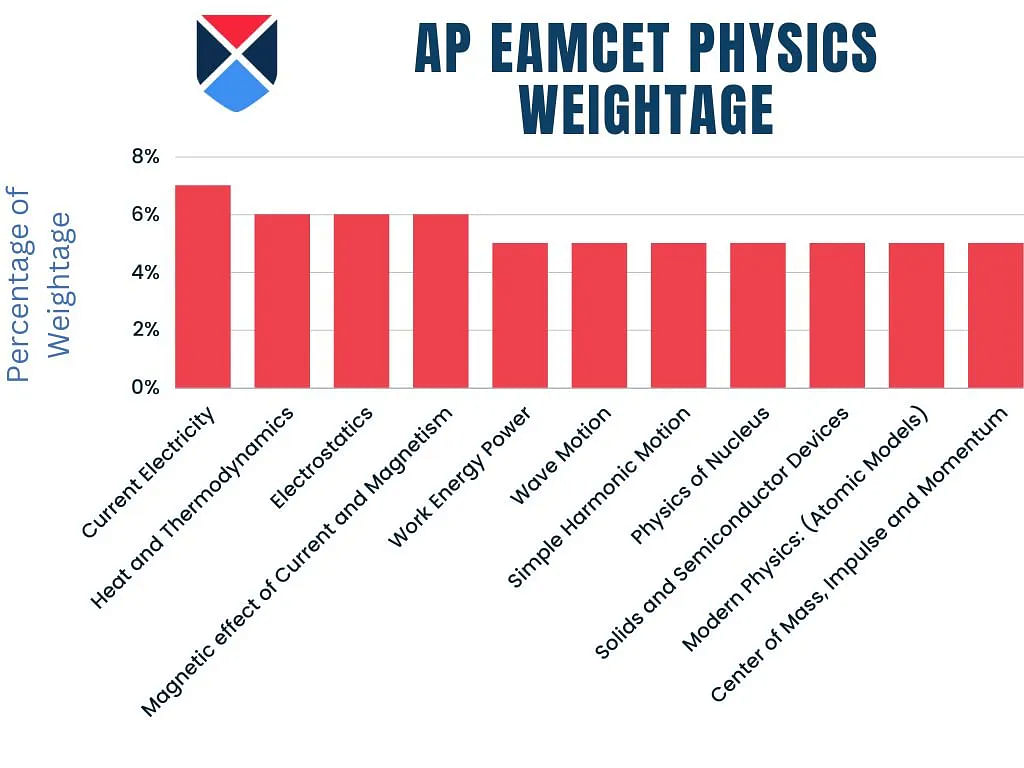
AP EAMCET Physics 2025 Topic Weightage
AP EAMCET topic-wise weightage of Physics for the year 2025 has been shared below for the candidates.
| Topic | Weightage |
| Current Electricity | 7% |
| Heat and Thermodynamics | 6% |
| Electrostatics | 6% |
| Magnetic effect of Current and Magnetism | 6% |
| Work Energy Power | 5% |
| Wave Motion | 5% |
| Simple Harmonic Motion | 5% |
| Physics of Nucleus | 5% |
| Solids and Semiconductor Devices | 5% |
| Modern Physics: (Atomic Models) | 5% |
| Center of Mass, Impulse and Momentum | 5% |
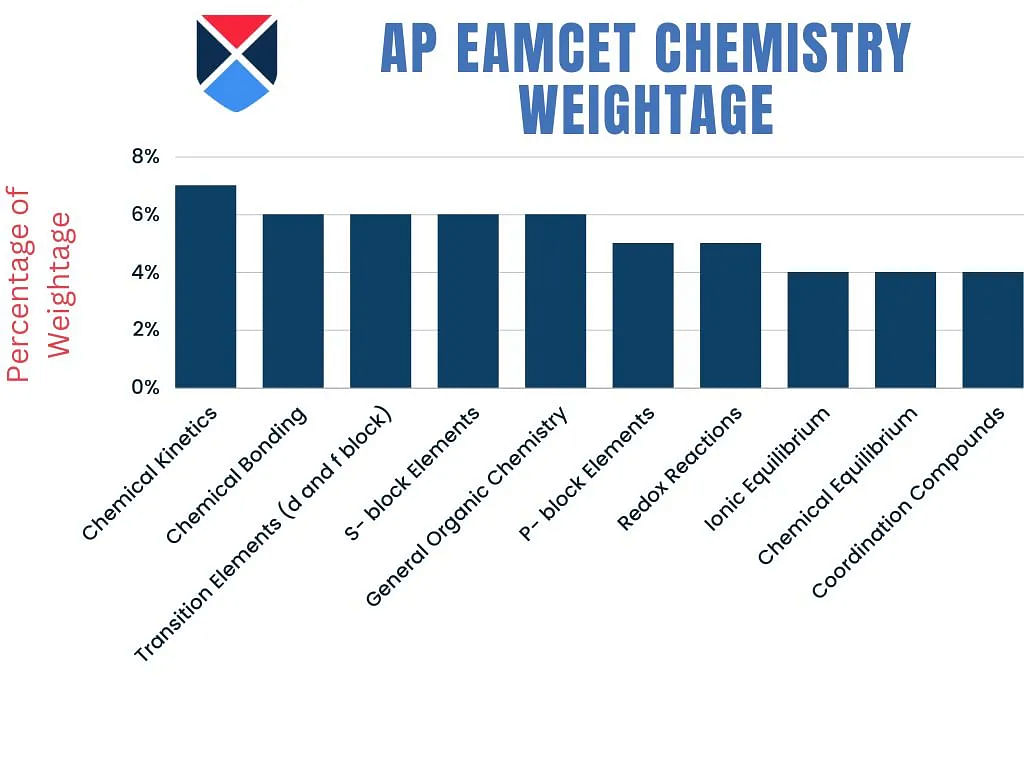
AP EAMCET Chemistry 2025 Topic Weightage
AP EAMCET topic-wise weightage of Chemistry for the year 2025 has been shared below for the candidates.
| Topic | Weightage |
| Chemical Kinetics | 7% |
| Chemical Bonding | 6% |
| Transition Elements (d and f block) | 6% |
| S- block Elements | 6% |
| General Organic Chemistry | 6% |
| P- block Elements | 5% |
| Redox Reactions | 5% |
| Ionic Equilibrium | 4% |
| Chemical Equilibrium | 4% |
| Coordination Compounds | 4% |
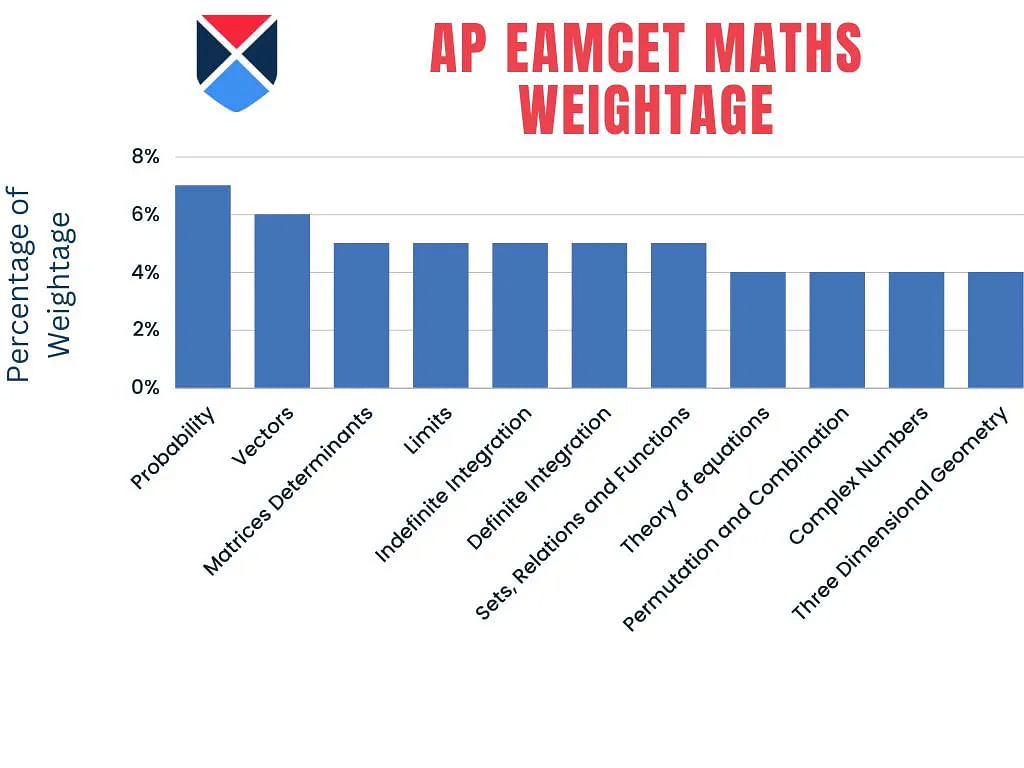
AP EAMCET Mathematics 2025 Topic Weightage
AP EAMCET topic-wise weightage of Mathematics for the year 2025 has been shared below for the candidates.
| Topic | Weightage |
| Probability | 7% |
| Vectors | 6% |
| Matrices Determinants | 5% |
| Limits | 5% |
| Indefinite Integration | 5% |
| Definite Integration | 5% |
| Sets, Relations and Functions | 5% |
| Theory of equations | 4% |
| Permutation and Combination | 4% |
| Complex Numbers | 4% |
| Three Dimensional Geometry | 4% |
AP EAMCET Exam Pattern 2025
The Jawaharlal Nehru Technological University, Kakinada, prescribes the exam pattern for AP EAMCET. Exam Pattern depends on several factors, such as the mode in which the exam will be conducted, question paper specification, exam duration, marking scheme, etc.
Therefore, candidates must have a brief knowledge of the EAMCET exam pattern BIPC appearing for the AP EAMCET 2025 examination. Candidates can have a look at the AP EAMCET exam pattern for 2025 below.
| Particulars | Details |
| Mode of Examination | Computer-Based Test (CBT) |
| Time Allotted for the Exam | 3 Hours |
| Language Medium | English and Telugu (Candidates who want to get questions translated in Urdu will be allotted a test centre at Kurnool only) |
| Section-wise Distribution of Questions | Physics- 40 QuestionsChemistry- 40 QuestionsMaths- 80 Questions |
| Format of Questions | Multiple-choice Questions |
| Total Number of Questions | 160 |
| Marking Scheme | 1 mark will be awarded for each correct answer. |
| Negative Marking | There is no negative marking for a wrong answer or unattempted questions. |
Knowing the exam pattern and syllabus is an advantage for the candidates so that they can properly prepare for the exam. The candidates need to prepare the correct syllabus for the AP EAMCET examination to succeed.
Read More: AP EAMCET Exam Pattern
AP EAMCET 2025 Reference Book
EAMCET AP syllabus reference books for all the courses for the year 2025 have been shared below. Candidates preparing for the AP EAMCET 2025 exam must check on the book names shared below.
Best AP EAMCET 2025 Mathematics Books
- Class XI & XII Mathematics -R.D. Sharma
- Problems Plus In IIT Mathematics- A. Das Gupta
- Trigonometry, Geometry Books- S.L. Loney
- Problems in Calculus of One Variable- I.A. Maron
- Handbook of Mathematics - A Multipurpose Quick Revision Resource- Arihant Experts
Best AP EAMCET 2025 Physics Books
- IIT JEE Physics- DC Pandey
- Concepts of Physics (Volume -1) -HC Verma
- Concepts of Physics (Volume- 2) -HC Verma
- EAMCET Physics (Andhra Pradesh & Telangana)- Arihant Experts
- Physics for IIT-JEE- Resnick, Halliday, Walker
Best AP EAMCET 2025 Chemistry Books
- Concise Inorganic Chemistry- J.D. Lee
- Organic Chemistry 7th Edition- Robert Thornton Morrison, Robert Neilson Boyd, Saibal Kanti Bhattacharjee
- EAMCET Chemistry Chapterwise 23 Years Solutions and 5 Mock Tests 3rd Edition- Arihant Experts
- Organic Chemistry- O.P Tandon
- Modern Approach to Chemical Calculations- R.C. Mukherjee
Read More:
FAQs on AP EAMCET Syllabus
Q: What is the step for the AP EAMCET 2024 syllabus PDF download?
Q: How many subjects are there in the EAMCET AP syllabus 2024?
Q: What is the syllabus for EAMCET 2024?
Q: Do NCERT books cover the whole AP EAMCET 2024 syllabus?
Q: What is the syllabus for Math for EAMCET 2024?